GEOMETRY OF PROTEINS
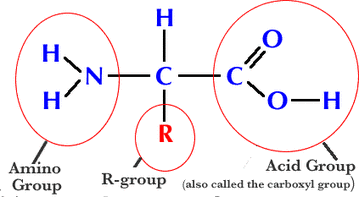
Protein molecules are made of amino acids which consist of the elements: oxygen, nitrogen, carbon, hydrogen, and sometimes sulphur. Protein can have anywhere between 50 and 5,000 amino acids in a single molecule. There are 20 types of amino acids in known existence, and they all have the same basic structure of:
The R-group stands for radical, which is an attachment of a variety of atoms. These atoms determine the the amino acid's properties and functions. An amino acid's elements that are bonded covalently are called the primary structure, while peptide bonds are what bond together two amino acids. Dehydration synthesis must take place in order for a peptide bond to be formed. Dehydration synthesis is when an O and OH group are removed among the two amino acids in order to give them the ability to form a peptide bond, and the result is the elimination of water and the bonding of two amino acids.